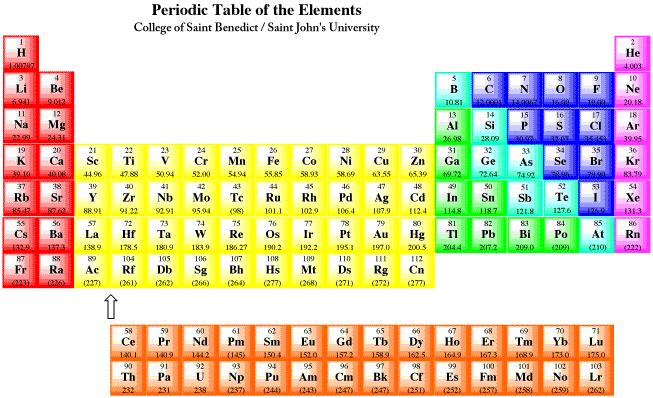
In particular, we focus on the similarities between elements in the same column and on the trends in properties that are observed across horizontal rows or down vertical columns. In this chapter, we explore the relationship between the electron configurations of the elements, as reflected in their arrangement in the periodic table, and their physical and chemical properties. Created by Sir William Crookes (1832–1919), the spiral represents the relationships between the elements and the order of evolution of the elements from what he believed to be primal matter. Chapter 6 "The Structure of Atoms" ended with the observation that, because all the elements in a column have the same valence electron configuration, the periodic table can be used to find the electron configuration of most of the elements at a glance.Ĭrookes’s Spiral Periodic Table, 1888. Thus because of their periodic similarities in electron configuration, atoms in the same column of the periodic table tend to form compounds with the same oxidation states and stoichiometries. 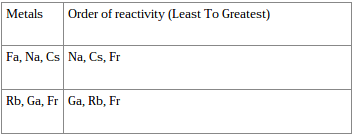
In contrast, the alkali metals have a single valence electron outside a closed shell and readily lose this electron to elements that require electrons to achieve an octet, such as the halogens. Because of their filled valence shells, the noble gases are generally unreactive. These closed shells are actually filled s and p subshells with a total of eight electrons, which are called octets helium is an exception, with a closed 1 s shell that has only two electrons. For example, the noble gases have what is often called filled or closed-shell valence electron configurations. In using this model to describe the electronic structures of the elements in order of increasing atomic number, we saw that periodic similarities in electron configuration correlate with periodic similarities in properties, which is the basis for the structure of the periodic table. In Chapter 6 "The Structure of Atoms", we presented the contemporary quantum mechanical model of the atom. To remember how the reactivity of the alkali metals and halogens increases or decreases, put a pin in the middle of the periodic table and spin it anti-clockwise.Chapter 7 The Periodic Table and Periodic Trends The outer shell will more easily attract another electron, which needs an electron to complete its full outer shell, when there is more attractive force.Ī useful mnemonic picture to help you recall that:Īs you go up group 7 (the halogens), again the elements get more reactive. The fewer electron shells (rings) between the nucleus and the outer shell (ring) also has less shielding effect and again this increases the electron attraction. 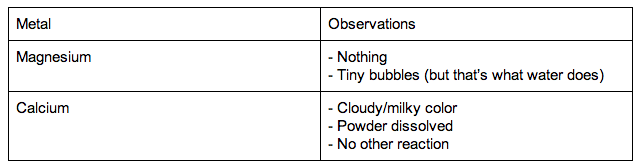
The distance "a" is less than "c" and the force of attraction between the nucleus and the outer shell increases with shorter distances. Halogens from bromide to fluorine get more reactive because the force of attraction between the nucleus (core) and the outer electron get stronger as you go up group 7 elements. Why do halogens get more reactive going upwards in group 7? The outer electron is more easily transferred to say an oxygen atom, which needs electrons to complete its full outer shell. The more electron shells (rings) between the nucleus and outer electron also creates shielding and again this weakens the nuclear attraction. The distance "c" is greater than "a" and the force of attraction between the nucleus and the outer shell (rings) diminishes with distance. Why do alkali metals get more reactive going down group 1?Īlkali metals from lithium to potassium get more reactive because the force of attraction between the nucleus (core) and the outer electron gets weaker as you go down group 1 elements. As you go down group 1 (the alkali metals) in the periodic table, the elements get more reactive.Īs you go up group 7 (the halogens), again, the elements become more reactive.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
